How Poor‑Quality Water Can Cause Eye Irritation When You Wear Contact Lenses
How contact lenses and water interact
When you expose lenses to tap water, dissolved minerals (calcium, magnesium; hardness often 60-180 mg/L), trace contaminants, and microbes transfer from hands or water to the lens surface, altering optics and tear-film interaction. Even low-level bacteria or protozoa can be entrapped in deposits; CDC guidance warns against any water contact with lenses because biofilms and mineral films markedly increase infection and irritation risk.
Lens materials, porosity, and surface chemistry
When you choose between hydrogel and silicone-hydrogel lenses, note hydrogels (water content ~38-79%) have nanometer-scale pores that permit greater uptake of ions, proteins and microbes, while silicone-hydrogels trade bulk water for higher oxygen permeability and surface treatments. Surface charge and wettability dictate binding: porous, hydrophilic materials tend to attract more mineral and bacterial adherence, worsening comfort and contamination potential.
Water absorption, swelling, and deposit formation
When your lenses absorb water they typically swell (commonly a few percent up to ~15% volume change), altering fit and oxygen transmission; dissolved calcium and magnesium precipitate, and tear proteins denature on the matrix, forming deposits that trap microbes. If your taps are hard or your hands carry residues, deposits and biofilms form faster, reducing comfort and increasing infection risk.
Mechanistically, osmotic swelling and ion exchange let calcium and magnesium (often 20-200 mg/L in harder supplies) precipitate as carbonate or phosphate onto your lens; increased ionic strength and neutral-to-basic pH accelerate protein aggregation and lipid binding. Clinical reports link showering or topping-up lenses with tap water to Acanthamoeba contamination, and lab studies show mineral-protein films can measurably reduce oxygen transmissibility (often ~5-20%) and shelter bacteria from disinfectants, amplifying irritation and risk.

Key Takeaways:
- Minerals and chemicals in tap water (calcium, magnesium, chlorine) can deposit on contact lenses, creating films that blur vision and irritate the ocular surface.
- Bacteria and protozoa, including Acanthamoeba, can transfer from hands or water to lens surfaces, adhere and multiply, raising the risk of painful infections and corneal inflammation.
- Household contaminants (soap residue, cosmetics, heavy metals, disinfectant byproducts) can transfer onto lenses and trigger allergic or inflammatory reactions; using sterile solutions and avoiding tap water contact reduces this risk.
Tap water components that can cause problems
Dissolved minerals, metals, and chemical residues
Hardness minerals like calcium and magnesium (water hardness >120 mg/L as CaCO3 is considered hard) can leave visible deposits and make your lenses feel gritty; metals such as lead (action level 15 ppb) and copper (1.3 mg/L) may transfer from your hands to lenses and irritate the cornea. Trace chemical residues from pesticides or pharmaceuticals (ng-µg/L) can adsorb to lens surfaces, alter the tear film, and increase protein buildup, raising infection and discomfort risk.
Chlorine, disinfectant byproducts, and organics
Municipal chlorine residuals typically range from 0.2-4.0 mg/L (EPA MRDL 4.0 mg/L) and can strip lipids from your tear film, causing immediate stinging and increased evaporation; disinfectant byproducts like total trihalomethanes (TTMs, EPA limit 80 ppb) and haloacetic acids can adsorb onto lenses and prolong irritation. Organic contaminants-volatile organics or algal toxins at µg/L levels-may also adhere to soft or silicone-hydrogel materials, worsening redness and discomfort.
Mechanistically, chlorine oxidizes proteins and lipids on both your lens and ocular surface, reducing tear stability and promoting microabrasions that let bacteria colonize more easily; disinfectant byproducts and organics can then act as irritants or reservoirs that slowly release into your eye. Practical examples include stinging after showering or washing hands before handling lenses-residual chlorine around 0.5 mg/L can be enough to trigger symptoms, and cumulative adsorption raises the chance of chronic irritation.
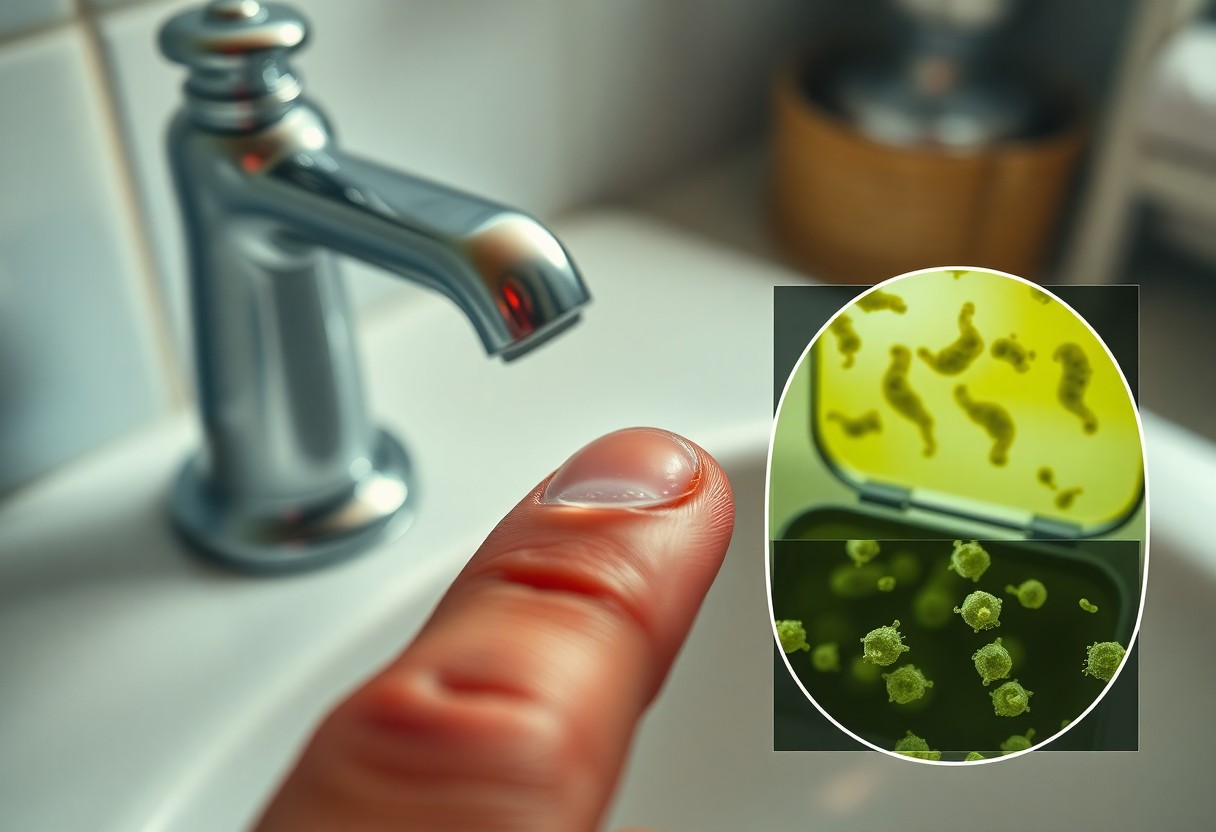
Microbial hazards in household water
Household water can carry organisms and particles that transfer from your hands to lenses, where minerals and proteins on the lens surface trap them against the cornea. Acanthamoeba, Pseudomonas and fungi like Fusarium have all been linked to contact‑lens keratitis; Acanthamoeba keratitis, though rare (~1-2 cases per million wearers annually), can cause severe vision loss, while Pseudomonas accounts for up to 50% of contact lens-related microbial keratitis in many studies.
Free‑living organisms (Acanthamoeba) and bacteria
Acanthamoeba lives in tap water, showerheads and hot tubs and forms hardy cysts that cling to lenses when you wet or touch them, leading to painful ulcers. Pseudomonas thrives in warm, nutrient‑rich water and can replicate rapidly (doubling times under optimal conditions), so when your hands contact faucet spray or sink biofilms you can transfer high loads of pathogenic bacteria directly to the lens surface.
Biofilms, fungi, and opportunistic pathogens
Biofilms coat pipes, faucet aerators and poorly cleaned lens cases, sheltering fungi like Fusarium, yeasts and opportunists such as Serratia marcescens; these communities intermittently shed cells into tap water, so when you touch a lens you may deposit a mix of organisms and organic debris. Biofilms protect pathogens from disinfectants, and past Fusarium outbreaks (hundreds of cases worldwide in 2005-2006) show how devastating lens‑associated fungal infections can be.
Minerals (calcium, magnesium) and organic films from tear proteins accelerate biofilm formation on lens surfaces, and biofilm‑embedded microbes can be tens to thousands of times more tolerant of disinfectants than free cells. That means low levels of chlorine in tap water often don’t neutralize organisms clinging to plumbing, so when your fingertips contact lenses you can transfer concentrated microbial communities-sometimes millions of cells per cm²-raising the risk of rapid, sight‑threatening infections.
How contaminants transfer from hands to lenses
When you handle lenses with damp or recently washed hands, microscopic droplets carry minerals (calcium, magnesium), bacteria such as Acanthamoeba and Pseudomonas, and particulate residues directly onto the lens surface; even brief contact deposits enough material to irritate your eye or seed an infection. For more on tap water risks see The Danger of Using Tap Water with Contact Lenses. Always dry hands thoroughly before touching lenses to reduce transfer.
Sources of hand contamination and inadequate drying
Faucet spray, sink biofilm, wet towels, lotions, makeup and food residues all sit on your hands and transfer to lenses when you touch them; swimming or showering before handling lenses increases exposure. Inadequate drying leaves microscopic water films and lint that trap minerals and microbes, so if you don’t pat your hands completely dry with a lint‑free towel, you raise the chance of deposits and contamination.
Adhesion mechanisms: oils, proteins, and particulate residues
Skin oils (sebum), makeup lipids and tear proteins like lysozyme rapidly coat lenses, creating a conditioning film that binds particulate minerals and microbes; those hydrophobic and electrostatic interactions make residues tough to remove and accelerate discomfort and infection risk.
More specifically, oils adhere via hydrophobic attraction while proteins unfold and expose sticky sites that trap minerals and cells; particles like calcium form crystalline deposits that scatter light and abrade the cornea. Lens material matters: hydrogel polymers tend to accumulate more proteins, whereas silicone hydrogels attract more lipids. Bacteria can exploit these films to form a biofilm within 24-48 hours, which substantially reduces disinfection efficacy and increases the likelihood of persistent infection if you transfer residues from your hands to the lens.

How transferred substances irritate the eye
When you touch lenses with hands that have minerals, bacteria or contaminants from tap water, those substances transfer and adhere to the lens surface. Hard-water minerals (TDS often 50-500 mg/L) form deposits that roughen the lens and change wettability, while residual chlorine, metals or soaps leave chemical irritants. Bacteria in household water (often hundreds to thousands of CFU/mL) can hitch a ride and become trapped against your cornea, causing reduced oxygen transmission, persistent irritation, and raised infection risk.
Mechanical and chemical irritation, allergic responses
Mineral flakes and particulates create microscopic abrasions on your corneal epithelium and disrupt lens movement, producing foreign‑body sensation and tearing. Chemical residues such as chlorinated disinfectants or metal ions can cause epithelial cell toxicity and sting on contact. Protein and particulate buildup also promotes mast‑cell activation, so you may develop contact lens‑associated allergic conjunctivitis with itching, redness and reduced comfort; micro‑abrasions and epithelial toxicity amplify these reactions.
Infection risk, keratitis, and inflammatory complications
Pathogens like Pseudomonas aeruginosa and Acanthamoeba often found in tap water can transfer to lenses and seed the cornea. Pseudomonas can produce rapid, ulcerative keratitis within 24-48 hours, while Acanthamoeba causes a slower, intensely painful, treatment‑resistant keratitis. Both can trigger intense inflammation, scarring, and vision loss if not treated promptly, making these organisms particularly sight‑threatening.
Clinically, infection signs include pain out of proportion to exam, decreased vision, photophobia and persistent discharge; Acanthamoeba cases frequently require months (commonly 3-6 months) of topical anti‑amoebic therapy and sometimes surgical intervention. Delayed diagnosis often leads to corneal scarring or the need for keratoplasty. Since simple contact with contaminated water can introduce these pathogens, the consequences range from prolonged therapy to permanent vision impairment if you don’t act quickly.
Minimizing risk: safe handling and alternatives to tap water
To lower risk, treat tap water as a contaminant: Contact Lenses and Water – say NO! to H2O explains why. When you rinse or wet lenses with tap water you can transfer minerals (calcium, magnesium), microbes like Pseudomonas and Acanthamoeba, and chlorine byproducts that cause chemical irritation, deposits, or sight‑threatening infections. Use sterile saline or FDA‑approved multipurpose or 3% hydrogen peroxide systems instead and avoid showering or swimming while wearing lenses.
Proper handwashing, drying, and handling technique
Wash your hands with soap and water for at least 20 seconds before handling lenses, rinse thoroughly, and dry with a clean, lint‑free towel. Use your fingertips to handle lenses, avoid contact with nails, and turn off the tap or use a paper towel to touch the faucet to prevent recontamination. If your hands touch tap water after washing, rewash-do not let water contact the lens surface.
Recommended cleaning solutions, storage, and when to avoid water
Choose FDA‑approved multipurpose disinfecting solutions or a neutralizing 3% hydrogen peroxide system; saline alone won’t disinfect. Rub and rinse lenses with solution for at least 10 seconds, replace your lens case every three months, and never “top off” old solution. Avoid using tap water or homemade saline and remove lenses before any water exposure (showers, pools, hot tubs) to reduce microbial contamination and deposit buildup.
Hydrogen peroxide systems must complete the full neutralization cycle-typically about 6 hours in the special case-or residual peroxide can irritate or burn your eye. Multipurpose solutions vary: some target protein removal while others focus on broad disinfection, so follow product labeling and your practitioner’s recommendation. Store cases upright to air dry at room temperature, discard any lens or solution exposed to tap water, and seek eye care promptly for persistent redness, pain, or blurred vision.
FAQ
Q: How can minerals and dissolved salts in tap water cause eye irritation when they get onto contact lenses?
A: Minerals and dissolved salts (calcium, magnesium, iron, bicarbonates) in tap water can deposit on the lens surface or on lenses handled with wet hands. Those deposits change a lens’s surface wettability and smoothness, increasing friction between the lens and the eyelid or cornea. Mineral films also scatter light and can create microscopic abrasions on the ocular surface, provoking stinging, redness, tearing and a gritty sensation. In addition, changes in local osmolarity and pH from residual tap water can draw fluid out of corneal cells or irritate tear-film balance, amplifying discomfort and making the eye more vulnerable to infection.
Q: Can bacteria and other microbes in poor‑quality tap water transfer to contact lenses and harm my eyes?
A: Yes. Tap water can contain bacteria (including Pseudomonas and other gram‑negative organisms), fungi and free‑living amoebae such as Acanthamoeba. When wet hands or water are used with lenses or lens cases, microbes adhere to the lens surface and can form biofilms that resist routine cleaning. Once attached, microbes can multiply and release toxins or invade the corneal surface, causing inflammation (conjunctivitis, keratitis) and severe pain, blurred vision or even sight‑threatening ulcers. Symptoms often start subtly but can progress rapidly, so early recognition and prompt care are important.
Q: What other contaminants in tap water cause eye irritation when transferred to lenses, and how can these be avoided?
A: Chemical contaminants – chlorine, chloramines, heavy metals (lead, copper), residual disinfectants, detergents from handwashing, and organic pollutants – can adhere to lenses after contact with tap water. These irritants produce toxic or hypersensitivity reactions, disrupt the tear film, and trigger redness, burning, itching or swelling. To avoid these problems, do not rinse or store contact lenses in tap water or handle lenses with wet hands; use sterile, manufacturer‑recommended contact solutions, dry hands thoroughly before handling lenses, avoid showering or swimming while wearing lenses, and replace lens cases and solution regularly to prevent contamination.
